Hemorphins are hemoglobin β-chain-derived peptides initially recognized for his or her analgesic results by way of binding to the opioid receptors belonging to the household of G protein-coupled receptor (GPCR), in addition to their physiological motion on blood strain.
However, their molecular mechanisms within the regulation of blood strain should not absolutely understood. Studies have reported an antihypertensive motion by way of the inhibition of the angiotensin-converting enzyme, a key enzyme within the renin-angiotensin system.
In this research, we hypothesized that hemorphins may additionally goal angiotensin II (AngII) kind 1 receptor (AT1R) as a key GPCR within the renin-angiotensin system.
To examine this, we examined the consequences of LVV-hemorphin-7 on AT1R transiently expressed in human embryonic kidney (HEK293) cells utilizing bioluminescence resonance power switch (BRET) know-how for the evaluation of AT1R/Gαq coupling and β-arrestin 2 recruitment.
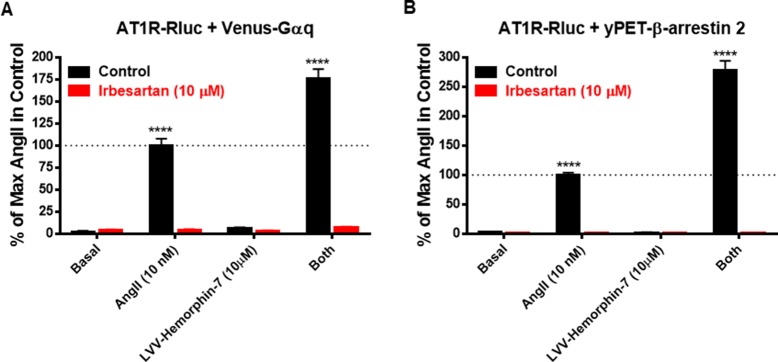
Interestingly, whereas LVV-hemorphin-7 alone had no vital impact on BRET alerts between AT1R and Gαq or β-arrestin 2, it properly potentiated AngII-induced BRET alerts and considerably elevated AngII efficiency.
The BRET information had been additionally correlated with AT1R downstream signing with LVV-hemorphin-7 potentiating the canonical AngII-mediated Gq-dependent inositol phosphate pathway in addition to the activation of the extracellular sign–regulated kinases (ERK1/2).
Both AngII and LVV-hemorphin-7-mediated responses had been absolutely abolished by AT1R antagonist demonstrating the focusing on of the lively conformation of AT1R. Our information report for the primary time the focusing on and the constructive modulation of AT1R signing by hemorphins, which can clarify their function within the physiology and pathophysiology of each vascular and renal techniques.
This discovering additional consolidates the pharmacological focusing on of GPCRs by hemorphins as beforehand proven for the opioid receptors in analgesia opening a brand new period for investigating the function of hemorphins in physiology and pathophysiology by way of the focusing on of GPCR pharmacology and signing.

Somewhat counterintuitively, the tyrosine phosphatase SHP-2 (SH2 domain-containing protein tyrosine phosphatase-2) is essential for the activation of extracellular sign–regulated kinase (ERK) downstream of varied progress issue receptors, thereby exerting important developmental capabilities.
This phosphatase additionally deploys proto-oncogenic capabilities and particular inhibitors have not too long ago been developed. With respect to the immune system, the function of SHP-2 within the signing of cytokines related for myelopoiesis and myeloid malignancies has been intensively studied.
The perform of this phosphatase downstream of cytokines essential for lymphocytes is much less understood, although a number of traces of proof counsel its significance. In addition, SHP-2 has been proposed to mediate the suppressive results of inhibitory receptors (IRs) that maintain a dysfunctional state in anticancer T cells.
Molecules concerned in IR signing are of potential pharmaceutical curiosity as blockade of these inhibitory circuits results in exceptional medical profit.
Here, we focus on the dichotomy within the capabilities ascribed to SHP-2 downstream of cytokine receptors and IRs, with a give attention to T and NK lymphocytes. Further, we spotlight the significance of broadening our understanding of SHP-2’s relevance in lymphocytes, an important step to tell on negative effects and unanticipated advantages of its therapeutic blockade.